For US Healthcare
Professionals Only
Learn more about the effectiveness of EMSAM®
Learn about EMSAM® dosing
Get information about the EMSAM® Savings Card
Efficacy Profile
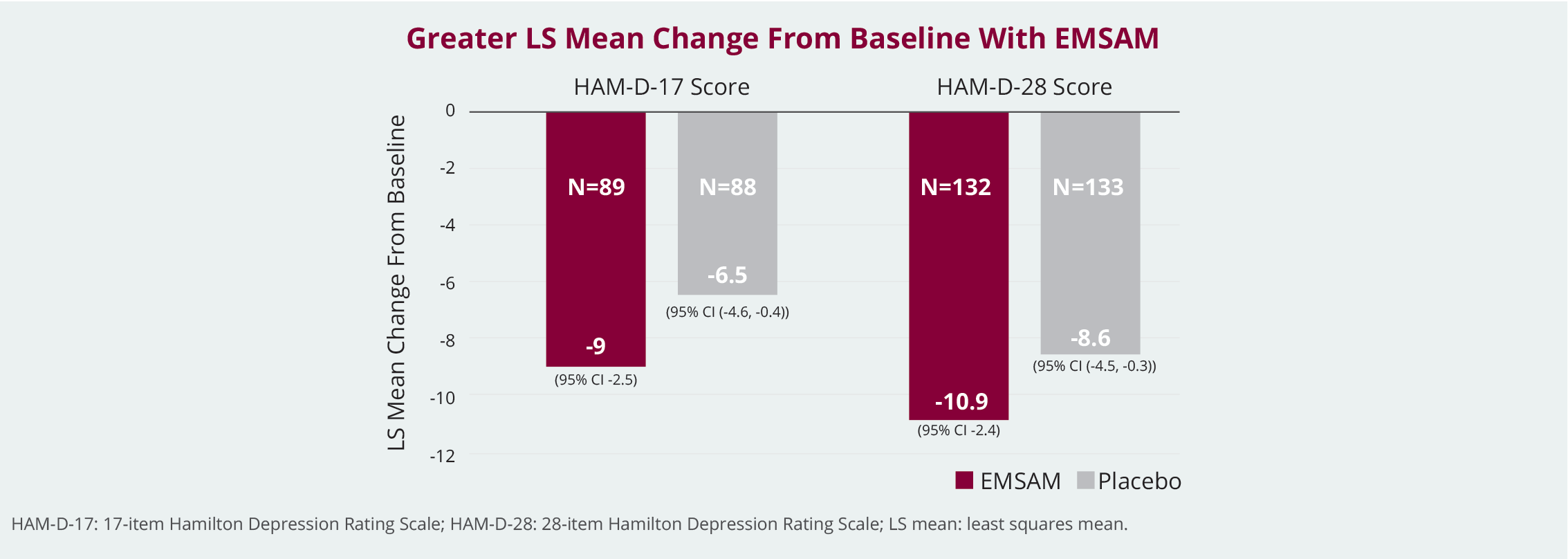
EMSAM® Demonstrated Better Clinical Efficacy Than Placebo in Adult Patients with MDD1*
EMSAM Significantly Improved MDD Symptoms Versus Placebo1*
EMSAM transdermal patch was more effective than placebo on the HAM-D scale.1*
*Study Descriptions1
The efficacy, safety, and tolerability of the selegiline transdermal system (EMSAM) were evaluated using 2 separate randomized, placebo-controlled, double-blind studies. The efficacy, safety, and tolerability of the selegiline transdermal system (EMSAM) were evaluated in outpatients aged 18 to 70 meeting DSM-IV criteria for Major Depressive Disorder using 2 separate randomized, placebo-controlled, double-blind studies. For study 1 (n = 89 for EMSAM and n = 88 for placebo), a 20 cm2 EMSAM patch (20 mg applied once daily) was evaluated over 6 weeks in adults with MDD. For study 2 (n = 132 for EMSAM and n = 133 for placebo), the dose range of 6 mg to 12 mg/24 hours was evaluated over 8 weeks. Primary efficacy outcomes used for evaluation were HAM-D Scale-17 for study 1 and HAM-D Scale-28 for study 2. In both studies, the EMSAM group showed significant improvement in the HAM-D total score (LS mean change from baseline of -9 for EMSAM vs -6.5 for placebo in Study 1 and -10.9 for EMSAM vs -8.6 for placebo in Study 2).
†Study Description
In another trial (study 3), the efficacy and safety of EMSAM (6 mg/24 h) were studied in a randomized, double-blind, placebo-controlled study in adult outpatients with MDD. In this 52-week study, 322 patients who responded with a 17-item HAM-D Scale score of 10 or less were randomly assigned at the same dose to EMSAM (n=159) or placebo (n=163). Discontinuation rates for each were at 52% for the EMSAM and placebo groups by week 12 of the double-blind phase. Patients who continued receiving EMSAM experienced significantly longer time to relapse compared to the placebo group patients.
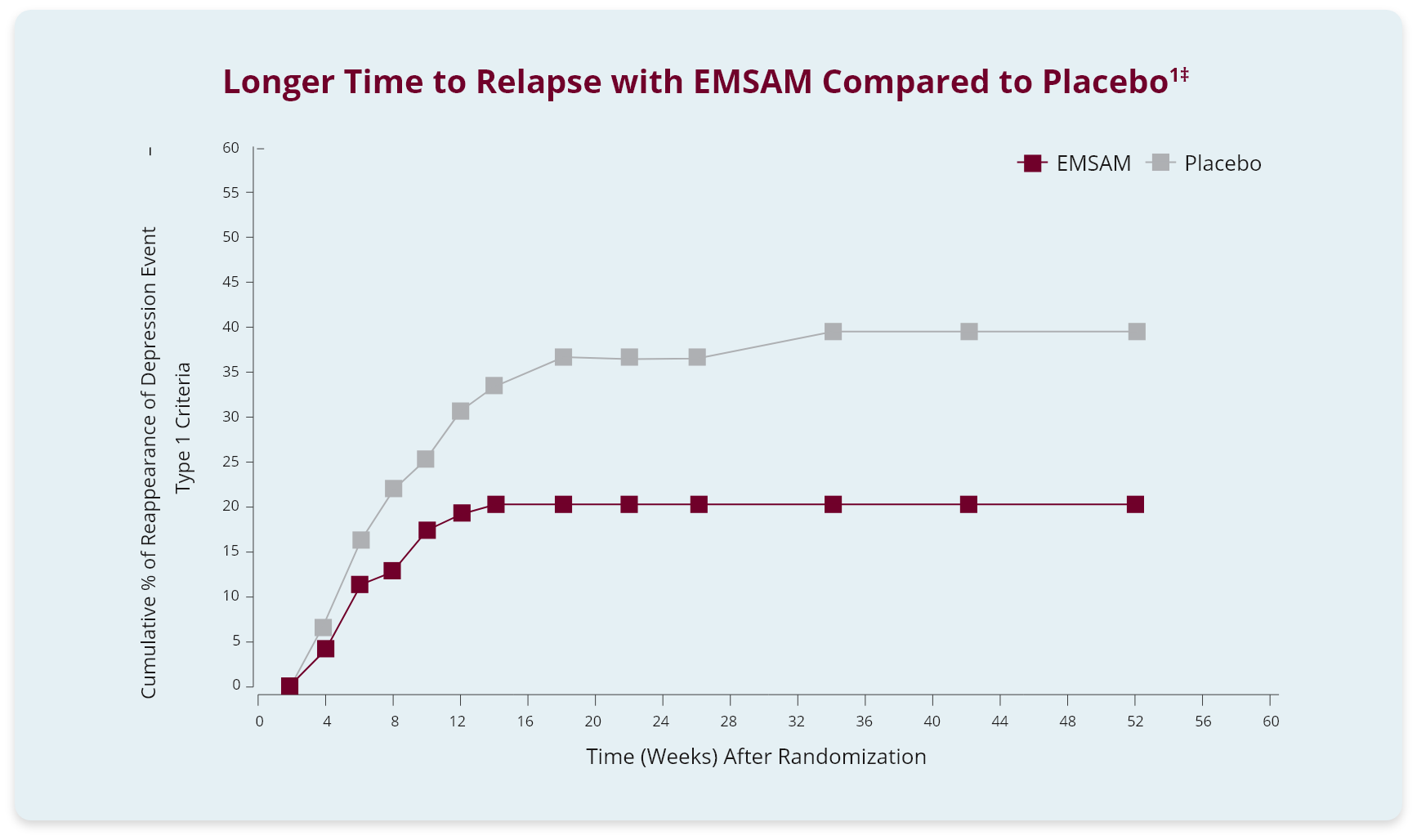
‡Relapse during the double-blind phase was defined as: (1) a 17-item HAM-D score of 14 or greater; (2) a CGI-S score of 3 or greater (with at least a 2-point increase from double-blind baseline); and (3) meeting DSM-IV criteria for MDD on 2 consecutive visits at least 11 days apart.
CGI-S: Clinical Global Impression-Severity Scale; MDD: Major Depressive Disorder.
Click here to learn more
about the adverse events
from these clinical studies
Your eligible commercially insured
patients may save up to $600*
*Eligibility restrictions apply. See full terms and conditions.
Safety Profile
Adverse Reactions: Incidence of ≥2% Among EMSAM®-Treated Patients.1
The Most Common Adverse Events Reported with EMSAM in Comparison to Placebo1*†
| Body System | EMSAM (N = 817) | Placebo (N = 668) | |
|---|---|---|---|
 |
Body as whole | ||
| Headache | 18 | 17 | |
 |
Digestive | ||
| Diarrhea | 9 | 7 | |
| Dyspepsia | 4 | 3 | |
 |
Nervous | ||
| Insomnia | 12 | 7 | |
| Dry mouth | 8 | 6 | |
 |
Respiratory | ||
| Pharyngitis | 3 | 2 | |
| Sinusitis | 3 | 1 | |
 |
Skin | ||
| Application site reaction | 24 | 12 | |
| Rash | 4 | 2 | |
*Excludes the following reactions, which had an incidence on placebo treatment greater or equal to EMSAM: infection, nausea, dizziness, pain, abdominal pain, nervousness, back pain, asthenia, anxiety, flu syndrome, accidental injury, somnolence, rhinitis, and palpitations.
- Includes doses of EMSAM from 3 mg to 12 mg per 24 hours in placebo-controlled trials of up to 8 weeks in duration.
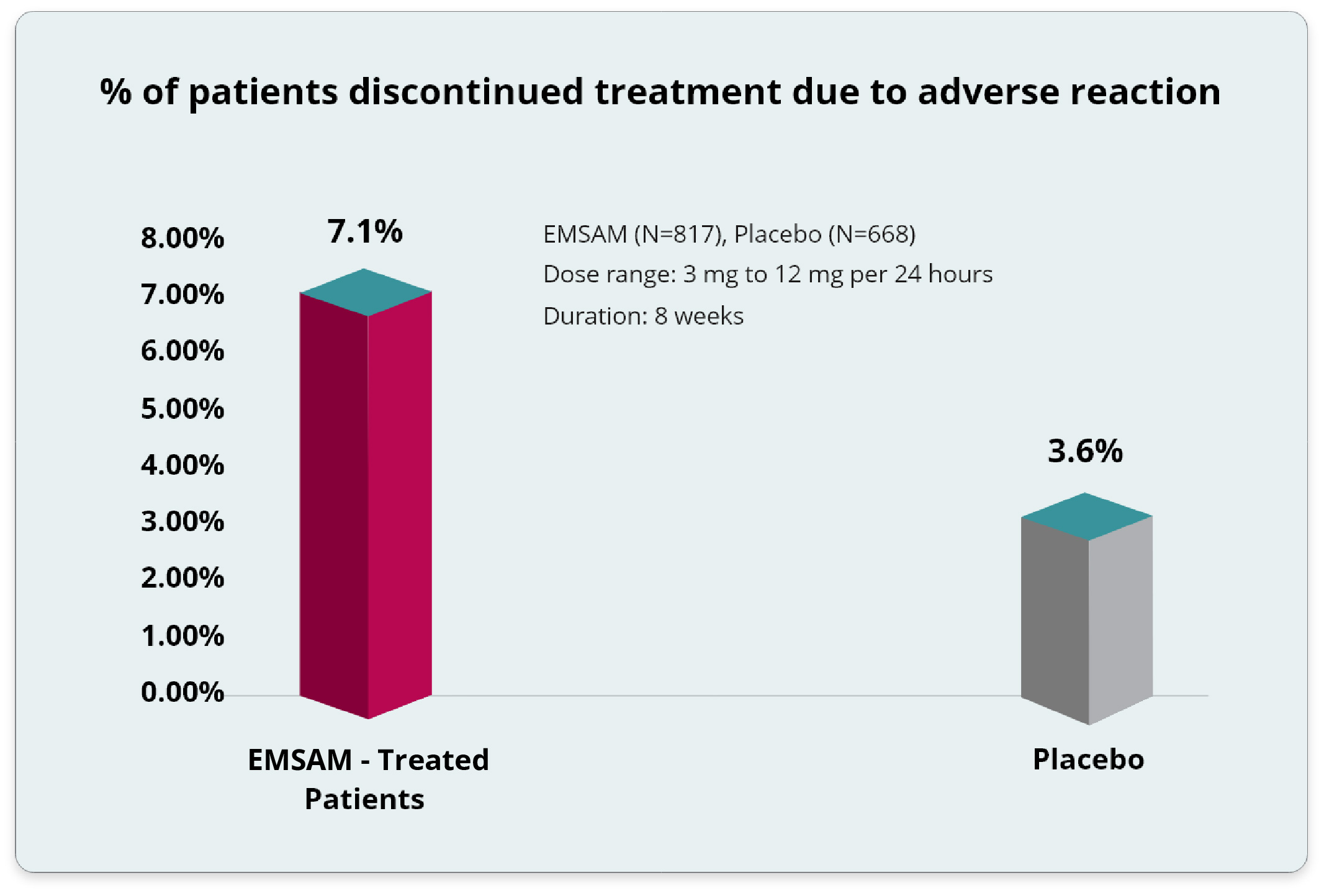
†Among N=817 MDD patients treated with EMSAM at doses of either 3 mg per 24 hours (n=151 patients), 6 mg per 24 hours (n=550 patients) or 6 mg per 24 hours, 9 mg per 24 hours, and 12 mg per 24 hours (n=116 patients) in placebo-controlled trials of up to 8 weeks in duration, 7.1% discontinued treatment due to an adverse reaction as compared with 3.6% of N=668 patients receiving placebo. The only adverse reaction associated with discontinuation, in at least 1% of EMSAM-treated patients at a rate at least twice that of placebo, was application site reaction (2% EMSAM vs. 0% placebo).

Sexual dysfunction
Incidence of side effects in EMSAM-treated patients was comparable to placebo in placebo-controlled trials. There are no adequately designed studies examining sexual dysfunction with EMSAM treatment.

Vital sign changes
EMSAM-treated patients experienced low systolic blood pressure compared to placebo. In a pool of short-term, placebo-controlled MDD studies, 3% of EMSAM patients and 1.5% of placebo patients experienced a low diastolic blood pressure (less than or equal to 90 mmHg with a change of baseline of at least 20 mmHg).
In one study with higher mean doses of EMSAM, 6.25% of EMSAM patients and 0% of placebo patients experienced a low standing systolic blood pressure by these criteria.
In the pool of short-term MDD trials, 9.8% of EMSAM-treated patients and 6.7% of placebo-treated patients experienced a notable orthostatic change in blood pressure, defined as a decrease of at least 10 mmHg in mean blood pressure with postural change.

Weight changes
Mean change in body weight: In placebo-controlled studies of 6-8 weeks duration, 2.1% of EMSAM patients (n=757) gained at least 5% compared to 2.4% of placebo patients (n=614). In these trials:
- Mean change in body weight for EMSAM-treated patients: 1.2 lbs loss
- Mean change in body weight for placebo-treated patients: 0.3 lbs gain

Laboratory and ECG changes
No clinically important changes were observed in various serum chemistry, hematology and urinalysis variables in the EMSAM vs placebo group.
No clinically significant changes in ECG parameters from baseline to final visit were observed in controlled studies with EMSAM (n=817) and placebo (n= 668) groups.

Other reactions observed during the premarketing evaluation of EMSAM
- Cardiovascular system: Tachycardia
- Digestive system: Anorexia
- Nervous system: Agitation, amnesia, tremor, twitching
- Skin and appendages: Pruritus
ECG: Electrocardiogram.
This is not all the information you need to review before prescribing EMSAM. Please also review the Important Safety Information below and the EMSAM Full Prescribing Information including Boxed WARNING.
Learn more about the
Efficacy Profile of EMSAM®
Your eligible commercially insured
patients may save up to $600*
*Eligibility restrictions apply. See full terms and conditions.
Dosage and Administration

EMSAM® Transdermal Patch Is Available in 3 Different
24-Hour Dosage Strengths1

- Initiate the treatment with EMSAM 6 mg/24 h dosage without any dietary restrictions1
- Avoid high tyramine food and beverages for 9 mg/24 h and 12 mg/24 h EMSAM doses1
- Maintenance of efficacy in depressed patients on therapy with EMSAM at a dose of 6 mg per 24 hours after achieving a responder status for an average duration of about 25 days was demonstrated in a controlled trial.
- The physician who elects to use EMSAM for extended periods should periodically re-evaluate the long-term usefulness of the drug for the individual patient.
- EMSAM 6 mg/24 h dosage requires no dietary restrictions!1
6 mg/24 h
Recommended starting dose and target dose

No dietary restrictions
9 mg/24 h OR 12 mg/24 h
Based on clinical judgment, dose can be increased in increments of 3 mg/24 h at intervals of no less than 2 weeks up to a maximum dose of 12 mg/24 h.* However, trials were not designed to assess whether higher doses are more effective than the lowest effective dose of 6 mg/24 h.
*Full antidepressant effect may be delayed.

For 9 mg/24 h and 12 mg/24 h doses, advise patients from the first dose of treatment to avoid high-tyramine foods and beverages. Continue avoiding up to a period of 2 weeks after a dose reduction to 6 mg/24 h or following the discontinuation of
9 mg/24 h or 12 mg/24 h dose.
Click here to learn more about tyramine-rich foods and
beverages to avoid.
Maintenance Treatment1
Administration of EMSAM®1
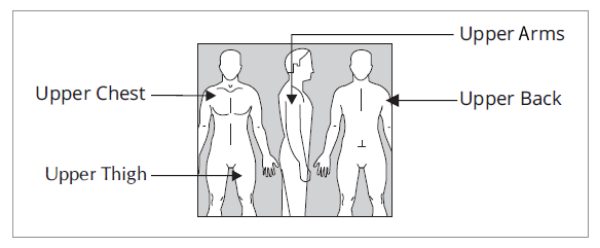
A new application site should be selected with each new transdermal system.
Where to apply?
Dry, intact skin on the upper torso (below the neck and above the waist), upper thigh or outer surface of the upper arm. Clothing and movement may make your patch rub off.
How often?
Once every 24 hours
When to apply?
Apply at approximately the same time each day.
Click here to learn more about how to apply EMSAM.
Systemic Absorption1
- 25% to 30% of the selegiline content on average is delivered systemically over 24 hours (range approximately 10% to 40%). Consequently, the degree of drug absorption may be 1/3rd higher than the average amounts of 6 mg to 12 mg per 24 hours.
- Transdermal dosing results in significantly higher exposure to selegiline with significantly lower exposure for all metabolites. Following dermal application of radio-labeled selegiline to laboratory animals, selegiline is rapidly distributed to all body tissues. Selegiline rapidly penetrates the blood-brain barrier. In humans, selegiline is approximately 90% bound to plasma protein over a 2 to 500 ng per mL concentration range. Selegiline does not accumulate in the skin.
Learn more about the
Efficacy Profile of EMSAM®
Your eligible commercially insured
patients may save up to $600*
*Eligibility restrictions apply. See full terms and conditions.
Savings Card Program
Your Eligible, Commercially Insured Patients May Be Able to Save on Their Out-of-Pocket Costs With the EMSAM® Savings Card*
EMSAM Savings Card – Save up
to $600 on EMSAM*
EMSAM, the first and only once-daily skin patch for the treatment of adults with MDD,1 is offering a Savings Card.
MDD: Major Depressive Disorder.

EMSAM® (selegiline transdermal system) Savings Card
Eligible, commercially insured patients may have their commercial copay amount for EMSAM® (selegiline transdermal system) reduced up to a maximum of $600 per month after the patient pays the first $20 per 30-day prescription, up to a maximum of $7,200 per calendar year.*
*Help your eligible patients to register by clicking here to get the Savings Card and to see full terms and conditions. Valid prescription with Prescriber ID# is required. Viatris Specialty LLC reserves the right to amend or end this program at any time without notice.
Learn more about the
Efficacy Profile of EMSAM®
Learn more about the Dosage
options available for EMSAM®
Patient Case Studies
Getting Karen started on EMSAM
- Karen, a 32-year-old female,* was diagnosed with MDD 2 years ago
- Patient was previously treated with first-line antidepressants
- Patient was looking for non-oral delivery, and the doctor recommends EMSAM
- Doctor has confirmed that the patient is not currently taking any serotonergic drugs
- Doctor waits 1 week from discontinuation of existing therapy before initiation of EMSAM to reduce the risk of serotonin syndrome
- The patient was initiated on a once-daily skin patch
– 6 mg/24 h - Based on the doctor's clinical judgment, the doctor increased Karen's dose to 9 mg/24 h. Dose increase should occur in increments of 3 mg/24 h and at intervals of no less than 2 weeks
- The doctor discusses dietary restrictions with the patient†
- EMSAM offers a potential MDD treatment option for adults who would prefer a non-oral therapy

*Hypothetical patient case study and dosing.
†For 9 mg/24 h or 12 mg/24 h EMSAM doses, patients are advised that they must follow a low-tyramine diet or avoid high-tyramine foods and beverages right from the first dose of treatment. Continue avoiding up to 2 weeks after a dose reduction to 6 mg/24 h or following the discontinuation of 9 mg/24 h or 12 mg/24 h dose.
Meet Mike
- Mike is a 50-year-old man,* who was diagnosed with Major Depressive Disorder (MDD)
- He has experienced 4 recurrences in last few years
- Mike was previously on an oral MAOI but wanted to try an alternative delivery option
- His doctor prescribed EMSAM as another treatment option for MDD.
Discuss your patient’s individual medical history and lifestyle, the safety and efficacy of the drug, dosing frequency and the dosage form to determine if EMSAM patch is right for them.
*Hypothetical patient case study.
Click here to learn more about patients who are not appropriate for EMSAM.

Getting Joseph started on EMSAM
- MAOIs like EMSAM in combination with a high-tyramine diet may cause a life-threatening hypertensive crisis
- The recommended starting and target dose of EMSAM is 6 mg per 24 hours. No dietary restrictions are required
- Based on clinical judgment, the dose of EMSAM can be increased in 3 mg increments in 2 week (or longer) intervals up to a maximum dose of 12 mg per 24 hours. Patients on 9 mg or 12 mg of EMSAM should avoid high-tyramine foods and beverages beginning on the first day of treatment at this dose until discontinuation. Learn more about the foods and beverages with high levels of tyramine that should be avoided